
 Copy to clipboard
Copy to clipboard 
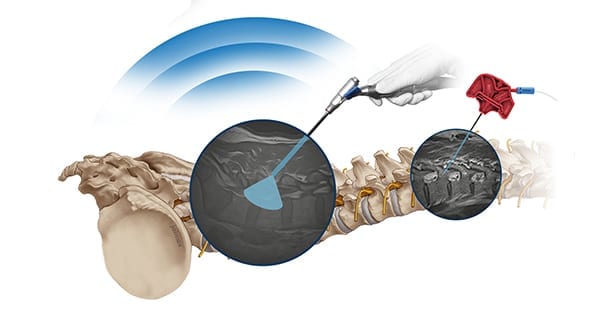
joimax received FDA 510(k) clearance to market Intracs®em, its electromagnetic navigation tracking and control system, for endoscopic spine surgery.
Intracs em allows spinal procedures to be carried out with minimal x-ray control, reducing radiation exposure, access time and length of the surgical procedure. All joimax instruments and scopes can be tracked and navigated through sensors. This improves safety, expands endoscopic indications and flattens the learning curve.
Intracs instruments are vector-guided at the tip, so the bending of needles or other pliant instruments will not affect navigational precision. The system can function as a stand-alone device or be integrated with the joimax Endoscopic Tower. It is reportedly the only system that allows for simultaneous navigation of multiple instruments such as needles, guiding rods, reamers and endoscopes. Intracs was launched in Europe in 4Q18.
Prof. Dr. med. Michael Kraus of ORTHIX Zentrum in Augsburg, Germany, who headed the preliminary trials, said, “Using Intracs®em Navigation System in the full endoscopic spine surgery is safe and easily applicable. The technology helps master precise access to the spine, and the clinical trials have confirmed the technique’s multiple advantages.”
joimax received FDA 510(k) clearance to market Intracs®em, its electromagnetic navigation tracking and control system, for endoscopic spine surgery.
Intracs em allows spinal procedures to be carried out with minimal x-ray control, reducing radiation exposure, access time and length of the surgical procedure. All joimax instruments and...
joimax received FDA 510(k) clearance to market Intracs®em, its electromagnetic navigation tracking and control system, for endoscopic spine surgery.
Intracs em allows spinal procedures to be carried out with minimal x-ray control, reducing radiation exposure, access time and length of the surgical procedure. All joimax instruments and scopes can be tracked and navigated through sensors. This improves safety, expands endoscopic indications and flattens the learning curve.
Intracs instruments are vector-guided at the tip, so the bending of needles or other pliant instruments will not affect navigational precision. The system can function as a stand-alone device or be integrated with the joimax Endoscopic Tower. It is reportedly the only system that allows for simultaneous navigation of multiple instruments such as needles, guiding rods, reamers and endoscopes. Intracs was launched in Europe in 4Q18.
Prof. Dr. med. Michael Kraus of ORTHIX Zentrum in Augsburg, Germany, who headed the preliminary trials, said, “Using Intracs®em Navigation System in the full endoscopic spine surgery is safe and easily applicable. The technology helps master precise access to the spine, and the clinical trials have confirmed the technique’s multiple advantages.”

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







