
 Copy to clipboard
Copy to clipboard 
Per results of AAOS’ last Orthopaedic Practice in the United States survey, a growing number of surgeons focus on specialties of extremities and sports medicine (or as we call it, Arthroscopy/Soft Tissue Repair). This year, for the first time at the AAOS annual meeting, new education specialty tracks are devoted to foot/ankle, shoulder/elbow, hand/wrist and sportsmed. What products were launched last year to serve these growing market segments? Let’s recap.
Select Extremities Joint Reconstruction Products Launched in 2017
ARTHROSURFACE
EU launch of an inlay glenoid component for OVO primary stemless total shoulder
- OVO is designed to restore the articular surface geometry of the humeral head and preserve with a 3-dimensional mapping system and contoured articular resurfacing implant
- As opposed to an onlay, the inlay is designed to significantly reduce glenoid loosening by eliminating the “rocking horse” effect
CORIN
U.S. launch of FX Solutions’ Humelock Reversed Shoulder
- Designed to treat massive rotator cuff tears and severe arthritis
- Device was FDA 510(k)-cleared in 1Q17 and offered in cemented and cementless designs
- Corin holds distribution rights for a variety of Humelock designs in Germany, the U.K. and the U.S.
DJO GLOBAL
AltiVate Anatomic Shoulder
- Features P2, a proprietary “porous porous” titanium coating designed to provide superior bone ingrowth
- Also features a short, bone-sparing humeral stem and a glenoid component with Drop and Go technology, encompassing trilobe features on the peripheral pegs for immediate fixation
EXTREMITY MEDICAL
Mid-carpal based total wrist arthroplasty system in the U.S. and select international markets (including the EU and Australia)
- Builds off of the clinical success of the KinematX Modular Hemi Wrist Arthroplasty System in the EU
FH ORTHOPEDICS
Broad U.S. launch of the Arrow Prime cementless glenoid baseplate
- Offers cementless humeral and glenoid platform solutions to address a variety of indications, including component loosening, and is accompanied by a universal instrument set
| INTEGRA LIFESCIENCES Cadence Total Ankle in Canada, the U.S. and select EU regions
Titan Press-Fit Reverse Shoulder for Fracture
|  Cadence Total Ankle |
LIMA CORPORATE
Celebrated the 15th anniversary of the SMR modular shoulder with new modular shoulder bone graft instruments and Lima Personalized Surgical Instruments with pre-op planning software
- SMR is reported to be the first shoulder platform with 15 years of clinical follow-up, and its modularity supports intra-op versatility and ease of conversion from anatomic to reverse, without removing the humeral stem and the Metal Back glenoid
- Portfolio includes anatomic, reverse, trauma, resurfacing and revision designs
| VIRTAMED ArthroS Ankle, reportedly the world’s first high-fidelity simulator for ankle arthroscopy
| 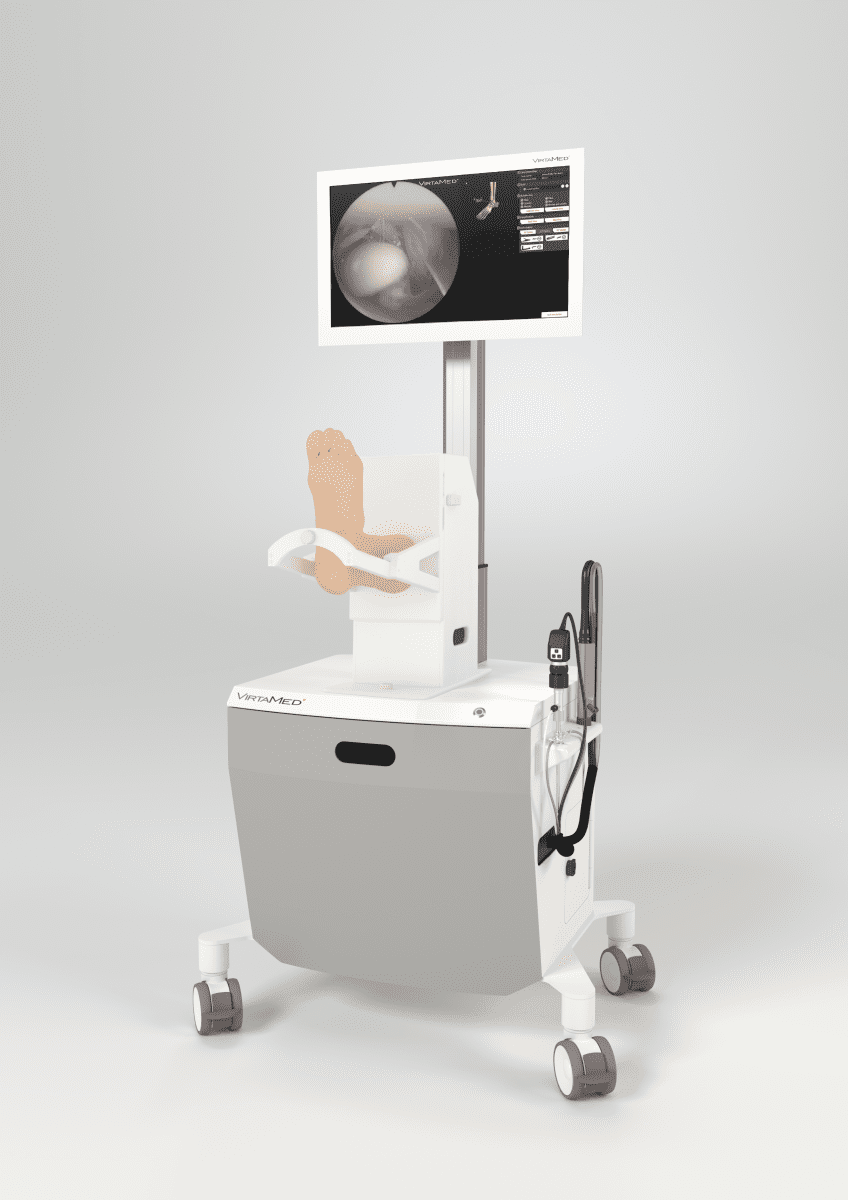 ArthoS Ankle Simulator |
Select Arthroscopy/Soft Tissue Repair Products Launched in 2017
AMPLITUDE SURGICAL
ACLip device for anterior cruciate ligament reconstruction
- During initial launch, device was implanted >850 times in an outpatient procedure
- Two-part design features a cage and endobutton, and instruments include a dual-level reamer that enables the drilling of two bone tunnels of different diameters in one procedure
- CE Mark approved in Europe; slated for U.S. FDA 510(k) submission
| DEPUY SYNTHES PUREVUE high-definition visualization system for minimally invasive endoscopic surgery
| 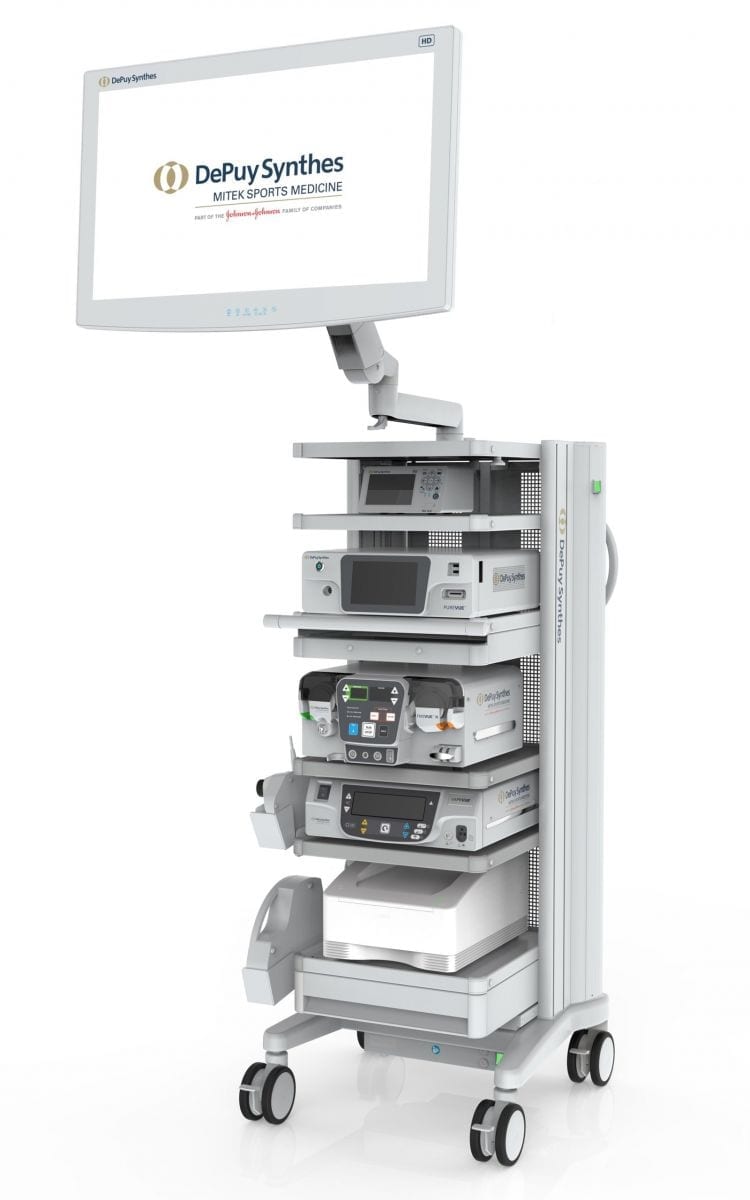 PUREVUE High-Def Visualization |
DEPUY SYNTHES MITEK SPORTS MEDICINE
Suite of knee arthroscopy implants and instruments for anterior cruciate ligament and meniscus repair
- SPEEDTRAP Graft Prep System for needle-free suturing in ~20 seconds (~77% faster than traditional techniques)
- TRUESPAN Meniscal Repair System offering one-handed operation and a 24-degree curved needle
- TWISTR Retrograde Reamer, for U.S. launch in 3Q17
- RIGIDLOOP Adjustable Cortical System
- INTRAFIX ADVANCE Tibial Fastener
DEPUY SYNTHES AND ETHICON
U.S. launch of wound closure kits customized for ortho-paedic surgery, including STRATAFIX Knotless Tissue Control Devices and the DERMABOND PRINEO Skin Closure System
- Designed to standardize and promote O.R. efficiency, enabling surgeons to choose sutures by each layer of closure
- Assembled and supplied by Medical Action Industries, an Owens & Minor Company that manufactures procedure-based kits and trays
ZIMMER BIOMET
International release of the Subchondroplasty procedure (SCP)
- Minimally invasive, outpatient procedure addresses defects associated with subchondral bone marrow lesions
- SCP fills the chronic lesions with AccuFill Bone Substitute, a porous injectable calcium phosphate, which is resorbed and replaced with healthy bone
Sources: Company press releases, websites, articles in the public domain; images courtesy of manufacturers
Julie A. Vetalice is the Editorial Assistant for ORTHOWORLD. Reach her by email.
Per results of AAOS’ last Orthopaedic Practice in the United States survey, a growing number of surgeons focus on specialties of extremities and sports medicine (or as we call it, Arthroscopy/Soft Tissue Repair). This year, for the first time at the AAOS annual meeting, new education specialty tracks are devoted to foot/ankle, shoulder/elbow,...
Per results of AAOS’ last Orthopaedic Practice in the United States survey, a growing number of surgeons focus on specialties of extremities and sports medicine (or as we call it, Arthroscopy/Soft Tissue Repair). This year, for the first time at the AAOS annual meeting, new education specialty tracks are devoted to foot/ankle, shoulder/elbow, hand/wrist and sportsmed. What products were launched last year to serve these growing market segments? Let’s recap.
Select Extremities Joint Reconstruction Products Launched in 2017
ARTHROSURFACE
EU launch of an inlay glenoid component for OVO primary stemless total shoulder
- OVO is designed to restore the articular surface geometry of the humeral head and preserve with a 3-dimensional mapping system and contoured articular resurfacing implant
- As opposed to an onlay, the inlay is designed to significantly reduce glenoid loosening by eliminating the “rocking horse” effect
CORIN
U.S. launch of FX Solutions’ Humelock Reversed Shoulder
- Designed to treat massive rotator cuff tears and severe arthritis
- Device was FDA 510(k)-cleared in 1Q17 and offered in cemented and cementless designs
- Corin holds distribution rights for a variety of Humelock designs in Germany, the U.K. and the U.S.
DJO GLOBAL
AltiVate Anatomic Shoulder
- Features P2, a proprietary “porous porous” titanium coating designed to provide superior bone ingrowth
- Also features a short, bone-sparing humeral stem and a glenoid component with Drop and Go technology, encompassing trilobe features on the peripheral pegs for immediate fixation
EXTREMITY MEDICAL
Mid-carpal based total wrist arthroplasty system in the U.S. and select international markets (including the EU and Australia)
- Builds off of the clinical success of the KinematX Modular Hemi Wrist Arthroplasty System in the EU
FH ORTHOPEDICS
Broad U.S. launch of the Arrow Prime cementless glenoid baseplate
- Offers cementless humeral and glenoid platform solutions to address a variety of indications, including component loosening, and is accompanied by a universal instrument set
| INTEGRA LIFESCIENCES Cadence Total Ankle in Canada, the U.S. and select EU regions
Titan Press-Fit Reverse Shoulder for Fracture
|  Cadence Total Ankle |
LIMA CORPORATE
Celebrated the 15th anniversary of the SMR modular shoulder with new modular shoulder bone graft instruments and Lima Personalized Surgical Instruments with pre-op planning software
- SMR is reported to be the first shoulder platform with 15 years of clinical follow-up, and its modularity supports intra-op versatility and ease of conversion from anatomic to reverse, without removing the humeral stem and the Metal Back glenoid
- Portfolio includes anatomic, reverse, trauma, resurfacing and revision designs
| VIRTAMED ArthroS Ankle, reportedly the world’s first high-fidelity simulator for ankle arthroscopy
|  ArthoS Ankle Simulator |
Select Arthroscopy/Soft Tissue Repair Products Launched in 2017
AMPLITUDE SURGICAL
ACLip device for anterior cruciate ligament reconstruction
- During initial launch, device was implanted >850 times in an outpatient procedure
- Two-part design features a cage and endobutton, and instruments include a dual-level reamer that enables the drilling of two bone tunnels of different diameters in one procedure
- CE Mark approved in Europe; slated for U.S. FDA 510(k) submission
| DEPUY SYNTHES PUREVUE high-definition visualization system for minimally invasive endoscopic surgery
|  PUREVUE High-Def Visualization |
DEPUY SYNTHES MITEK SPORTS MEDICINE
Suite of knee arthroscopy implants and instruments for anterior cruciate ligament and meniscus repair
- SPEEDTRAP Graft Prep System for needle-free suturing in ~20 seconds (~77% faster than traditional techniques)
- TRUESPAN Meniscal Repair System offering one-handed operation and a 24-degree curved needle
- TWISTR Retrograde Reamer, for U.S. launch in 3Q17
- RIGIDLOOP Adjustable Cortical System
- INTRAFIX ADVANCE Tibial Fastener
DEPUY SYNTHES AND ETHICON
U.S. launch of wound closure kits customized for ortho-paedic surgery, including STRATAFIX Knotless Tissue Control Devices and the DERMABOND PRINEO Skin Closure System
- Designed to standardize and promote O.R. efficiency, enabling surgeons to choose sutures by each layer of closure
- Assembled and supplied by Medical Action Industries, an Owens & Minor Company that manufactures procedure-based kits and trays
ZIMMER BIOMET
International release of the Subchondroplasty procedure (SCP)
- Minimally invasive, outpatient procedure addresses defects associated with subchondral bone marrow lesions
- SCP fills the chronic lesions with AccuFill Bone Substitute, a porous injectable calcium phosphate, which is resorbed and replaced with healthy bone
Sources: Company press releases, websites, articles in the public domain; images courtesy of manufacturers
Julie A. Vetalice is the Editorial Assistant for ORTHOWORLD. Reach her by email.

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







