Copy to clipboard
Copy to clipboard 
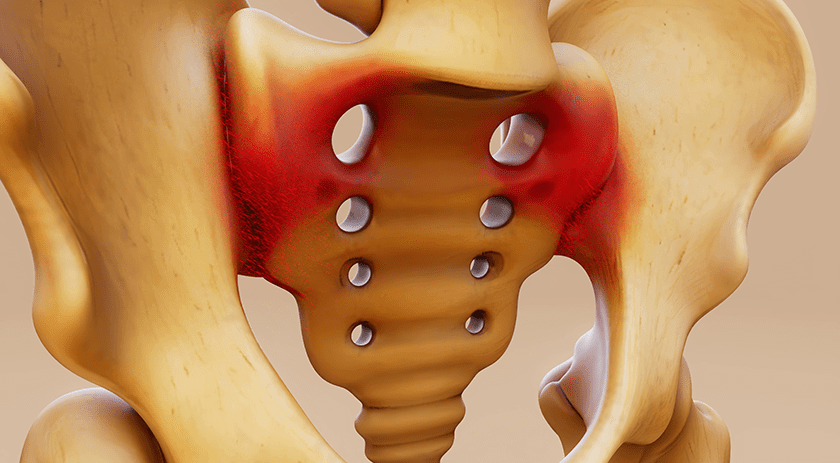
Exactech received FDA 510(k) clearance to market the Alteon® Acetabular Cup and XLE® Liner for hip reconstruction.
The cup provides multiple implant configurations and bearing options for the Alteon portfolio and features TAC™ asymmetric porous coating.
Alteon XLE highly crosslinked vitamin-E enhanced acetabular liners undergo a patented gamma irradiation and mechanical annealing process, to achieve the desired crosslink density.
In 2H19, Exactech will expand U.S. rollout of the Alteon Cup; full launch is slated for late 2019.
Source: Exactech

Exactech received FDA 510(k) clearance to market the Alteon® Acetabular Cup and XLE® Liner for hip reconstruction.
The cup provides multiple implant configurations and bearing options for the Alteon portfolio and features TAC™ asymmetric porous coating.
Alteon XLE highly crosslinked vitamin-E enhanced acetabular liners undergo a patented...
Exactech received FDA 510(k) clearance to market the Alteon® Acetabular Cup and XLE® Liner for hip reconstruction.
The cup provides multiple implant configurations and bearing options for the Alteon portfolio and features TAC™ asymmetric porous coating.
Alteon XLE highly crosslinked vitamin-E enhanced acetabular liners undergo a patented gamma irradiation and mechanical annealing process, to achieve the desired crosslink density.
In 2H19, Exactech will expand U.S. rollout of the Alteon Cup; full launch is slated for late 2019.
Source: Exactech


You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.