
 Copy to clipboard
Copy to clipboard 
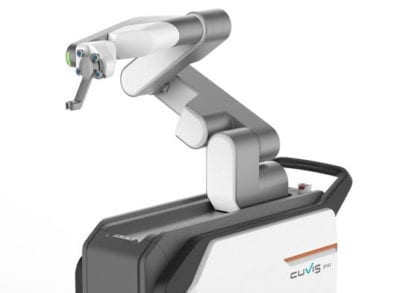
Curexo launched the CUVIS-spine robot-assisted spine surgery device following approval by Korea’s Ministry of Food and Drug Safety.
CUVIS-spine guides and supports the pedicle screw to the correct position using a robotic arm, one-step surgical tool and real-time location sensor. Real-time patient position monitoring can correct the surgical plan in real time, and can help reduce radiation exposure for the patient and O.R. staff.
Curexo seeks to establish an overseas sales agency, as it expects to receive CE Mark approval in 1H20 and will also apply for FDA clearance.
The company is also developing CUVIS-joint for launch in 2020.
Curexo launched the CUVIS-spine robot-assisted spine surgery device following approval by Korea's Ministry of Food and Drug Safety.
CUVIS-spine guides and supports the pedicle screw to the correct position using a robotic arm, one-step surgical tool and real-time location sensor. Real-time patient position monitoring can correct the...
Curexo launched the CUVIS-spine robot-assisted spine surgery device following approval by Korea’s Ministry of Food and Drug Safety.
CUVIS-spine guides and supports the pedicle screw to the correct position using a robotic arm, one-step surgical tool and real-time location sensor. Real-time patient position monitoring can correct the surgical plan in real time, and can help reduce radiation exposure for the patient and O.R. staff.
Curexo seeks to establish an overseas sales agency, as it expects to receive CE Mark approval in 1H20 and will also apply for FDA clearance.
The company is also developing CUVIS-joint for launch in 2020.

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







