
 Copy to clipboard
Copy to clipboard 
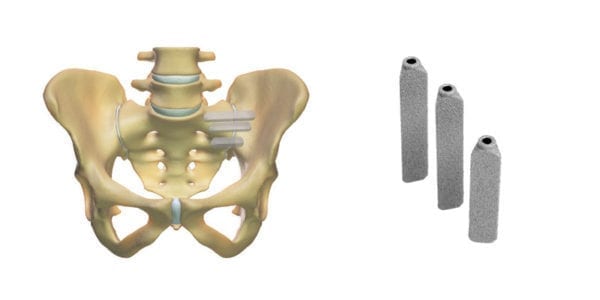
SI-BONE announced that Blue Cross Blue Shield Association (BCBSA) Evidence Street’s annual technology evidence review states that iFuse Transiliac Triangular Implants are the only implants for the treatment of sacroiliac joint pain with evidence sufficient to determine a meaningful improvement in the net health outcome. BCBSA has determined that the evidence is insufficient for any therapies or technologies that do not include transiliac placement of triangular implants.
Following this technology evidence review by BCBSA, Arkansas Blue Cross Blue Shield and BlueAdvantage Administrators of Arkansas, the plan administrator for Walmart employees, updated their coverage policies for minimally invasive SI joint fusion from non-coverage, to covering exclusively when transiliac triangular implants are used. Arkansas Blue Cross Blue Shield joins a majority of other BCBSA plans in establishing iFuse as the only product which may be used to treat patients with SI joint pain.
“In 2018, BCBSA first published its annual technology evidence review that singled out iFuse triangular implants as the only SI joint fusion implant with sufficient evidence to determine a meaningful improvement in patients’ net health outcome,” said Jeffrey Zigler, Vice President of Market Access and Reimbursement. “This updated evidence review further clarifies that only SI joint fusion performed through a transiliac approach and using iFuse triangular implants has sufficient evidence. BCBSA now distinguishes the evidence for iFuse from implants that are screw-based, or that are not placed through a transiliac approach.”
Source: SI-BONE, Inc.
SI-BONE announced that Blue Cross Blue Shield Association (BCBSA) Evidence Street’s annual technology evidence review states that iFuse Transiliac Triangular Implants are the only implants for the treatment of sacroiliac joint pain with evidence sufficient to determine a meaningful improvement in the net health outcome. BCBSA has determined that...
SI-BONE announced that Blue Cross Blue Shield Association (BCBSA) Evidence Street’s annual technology evidence review states that iFuse Transiliac Triangular Implants are the only implants for the treatment of sacroiliac joint pain with evidence sufficient to determine a meaningful improvement in the net health outcome. BCBSA has determined that the evidence is insufficient for any therapies or technologies that do not include transiliac placement of triangular implants.
Following this technology evidence review by BCBSA, Arkansas Blue Cross Blue Shield and BlueAdvantage Administrators of Arkansas, the plan administrator for Walmart employees, updated their coverage policies for minimally invasive SI joint fusion from non-coverage, to covering exclusively when transiliac triangular implants are used. Arkansas Blue Cross Blue Shield joins a majority of other BCBSA plans in establishing iFuse as the only product which may be used to treat patients with SI joint pain.
“In 2018, BCBSA first published its annual technology evidence review that singled out iFuse triangular implants as the only SI joint fusion implant with sufficient evidence to determine a meaningful improvement in patients’ net health outcome,” said Jeffrey Zigler, Vice President of Market Access and Reimbursement. “This updated evidence review further clarifies that only SI joint fusion performed through a transiliac approach and using iFuse triangular implants has sufficient evidence. BCBSA now distinguishes the evidence for iFuse from implants that are screw-based, or that are not placed through a transiliac approach.”
Source: SI-BONE, Inc.

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







