Copy to clipboard
Copy to clipboard 
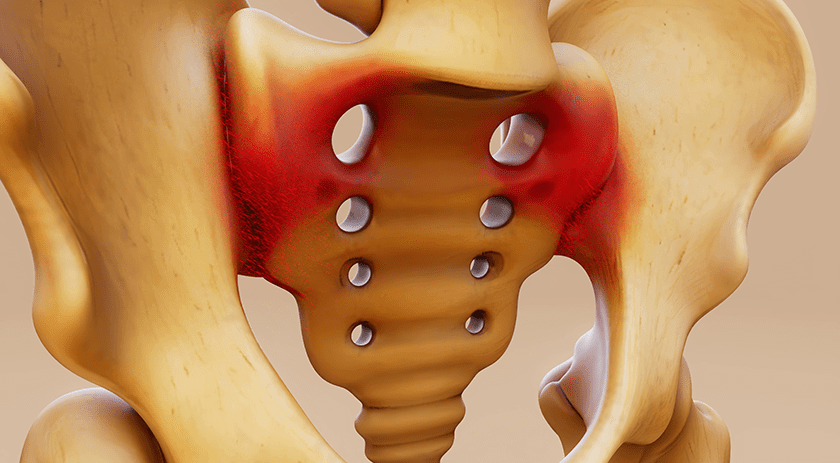
ChoiceSpine received FDA 510(k) clearance to market the HAWKEYE™ Ti 3D-printed titanium vertebral body replacement for use in the cervical spine, thus opening its use to all three spinal segments: cervical, thoracic and lumbar. Expansion of commercial launch will continue through 2Q19.
The HAWKEYE portfolio includes devices in a small or medium size footprint ranging from 14 mm to 60 mm in height, with a built-in 0° or 8° of lordosis. A large center chamber accepts bone graft, and lateral windows allow surgeons to assess spinal fusion over time.
Source: ChoiceSpine
ChoiceSpine received FDA 510(k) clearance to market the HAWKEYE™ Ti 3D-printed titanium vertebral body replacement for use in the cervical spine, thus opening its use to all three spinal segments: cervical, thoracic and lumbar. Expansion of commercial launch will continue through 2Q19.
The HAWKEYE portfolio includes devices in a small or...
ChoiceSpine received FDA 510(k) clearance to market the HAWKEYE™ Ti 3D-printed titanium vertebral body replacement for use in the cervical spine, thus opening its use to all three spinal segments: cervical, thoracic and lumbar. Expansion of commercial launch will continue through 2Q19.
The HAWKEYE portfolio includes devices in a small or medium size footprint ranging from 14 mm to 60 mm in height, with a built-in 0° or 8° of lordosis. A large center chamber accepts bone graft, and lateral windows allow surgeons to assess spinal fusion over time.
Source: ChoiceSpine

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.