
 Copy to clipboard
Copy to clipboard 
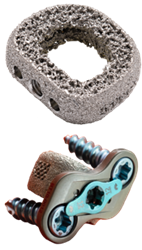
ChoiceSpine received FDA 510(k) clearance to market the TIGER SHARK™ C interbody device with BioBond™ technology and the BOOMERANG™ Anterior Cervical Plate (ACP), for use together or separately in cervical spinal fusion.
BOOMERANG instrumentation is designed to allow simultaneous insertion of the BOOMERANG plate with cervical interbodies, such as TIGER SHARK C or STEALTH™, mimicking a stand-alone ACDF concept and reducing O.R. time. The BOOMERANG ACP features a two-screw configuration and single-step screw blocking mechanism to optimize insertion and fixation.
TIGER SHARK C is a 3D-manufactured titanium alloy interbody created with BioBond organic, porous interbody architecture designed for continuous porosity with an osteoconductive, hydrophilic surface.
Source: ChoiceSpine
ChoiceSpine received FDA 510(k) clearance to market the TIGER SHARK™ C interbody device with BioBond™ technology and the BOOMERANG™ Anterior Cervical Plate (ACP), for use together or separately in cervical spinal fusion.
BOOMERANG instrumentation is designed to allow simultaneous insertion of the BOOMERANG plate with cervical interbodies, such...
ChoiceSpine received FDA 510(k) clearance to market the TIGER SHARK™ C interbody device with BioBond™ technology and the BOOMERANG™ Anterior Cervical Plate (ACP), for use together or separately in cervical spinal fusion.
BOOMERANG instrumentation is designed to allow simultaneous insertion of the BOOMERANG plate with cervical interbodies, such as TIGER SHARK C or STEALTH™, mimicking a stand-alone ACDF concept and reducing O.R. time. The BOOMERANG ACP features a two-screw configuration and single-step screw blocking mechanism to optimize insertion and fixation.
TIGER SHARK C is a 3D-manufactured titanium alloy interbody created with BioBond organic, porous interbody architecture designed for continuous porosity with an osteoconductive, hydrophilic surface.

Source: ChoiceSpine

You are out of free articles for this month
Subscribe as a Guest for $0 and unlock a total of 5 articles per month.
You are out of five articles for this month
Subscribe as an Executive Member for access to unlimited articles, THE ORTHOPAEDIC INDUSTRY ANNUAL REPORT and more.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







