Copy to clipboard
Copy to clipboard 
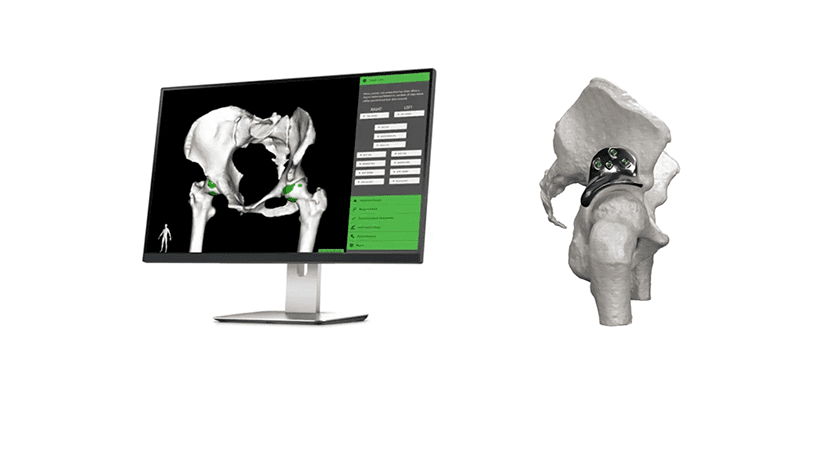
Bioventus invested US $2.5MM in the ongoing clinical study of CartiHeal’s Agili-C™ implant for the treatment of joint surface lesions. The infusion adds to a previous raise of $18.3MM and brings CartiHeal’s funding total to ~$21MM.
The Investigational Device Exemption trial kicked off in early 4Q17 with enrollment of 16 patients; since then, that number has risen over 30.
The multinational study will enroll 250 patients minimum with an objective to demonstrate the superiority of Agili-C vs. microfracture and debridement to treat cartilage or osteochondral defects.
Sources: CartiHeal and Bioventus; ORTHOWORLD Inc.
Bioventus invested US $2.5MM in the ongoing clinical study of CartiHeal's Agili-C™ implant for the treatment of joint surface lesions. The infusion adds to a previous raise of $18.3MM and brings CartiHeal's funding total to ~$21MM.
The Investigational Device Exemption trial kicked off in early 4Q17 with enrollment of 16 patients; since then,...
Bioventus invested US $2.5MM in the ongoing clinical study of CartiHeal’s Agili-C™ implant for the treatment of joint surface lesions. The infusion adds to a previous raise of $18.3MM and brings CartiHeal’s funding total to ~$21MM.
The Investigational Device Exemption trial kicked off in early 4Q17 with enrollment of 16 patients; since then, that number has risen over 30.
The multinational study will enroll 250 patients minimum with an objective to demonstrate the superiority of Agili-C vs. microfracture and debridement to treat cartilage or osteochondral defects.
Sources: CartiHeal and Bioventus; ORTHOWORLD Inc.

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.