
 Copy to clipboard
Copy to clipboard 
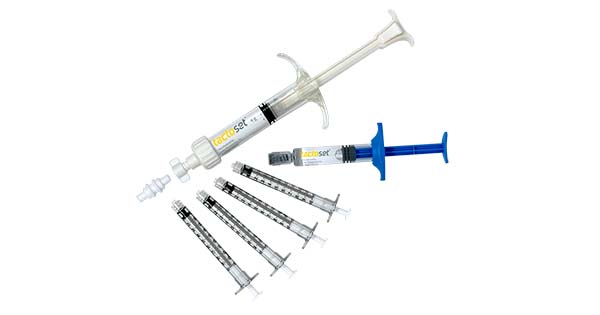
Anika Therapeutics received an additional FDA 510(k) clearance for Tactoset® Injectable Bone Substitute. This new indication expands the use of Tactoset to include augmentation of hardware and the support of bone fragments during surgical procedures. This expands Tactoset’s addressable market to include augmentation of suture anchor fixation in addition to treatment of skeletal system defects such as insufficiency fractures.
Tactoset is a calcium phosphate based, biocompatible bone graft substitute that incorporates Anika’s core hyaluronic acid (HA) technology. The HA component of Tactoset makes the product highly flowable, easily injectable and able to interdigitate trabecular bone architecture with overall improved handling characteristics compared to competitive products. Once injected, Tactoset hardens and mimics the properties of normal trabecular bone initially and remodels into healthy bone over time.
Since its launch in 4Q19, Tactoset has been used for the treatment of bone voids, insufficiency fractures and other skeletal defects, often performed in an outpatient surgical setting. With this expanded indication, surgeons can now use Tactoset in situations where augmenting hardware and/or bone fragments due to insufficient bone quality may be beneficial to their patients. Tactoset has been shown to increase the pull-out strength of a screw-in suture anchor two-fold1 in an osteoporotic bone model when augmented with the cured Tactoset compared to a suture anchor alone.
Anika expects to begin actively marketing the expanded indication for Tactoset for hardware augmentation in October 2021.
Source: Anika Therapeutics, Inc.
Anika Therapeutics received an additional FDA 510(k) clearance for Tactoset® Injectable Bone Substitute. This new indication expands the use of Tactoset to include augmentation of hardware and the support of bone fragments during surgical procedures. This expands Tactoset’s addressable market to include augmentation of suture anchor fixation in...
Anika Therapeutics received an additional FDA 510(k) clearance for Tactoset® Injectable Bone Substitute. This new indication expands the use of Tactoset to include augmentation of hardware and the support of bone fragments during surgical procedures. This expands Tactoset’s addressable market to include augmentation of suture anchor fixation in addition to treatment of skeletal system defects such as insufficiency fractures.
Tactoset is a calcium phosphate based, biocompatible bone graft substitute that incorporates Anika’s core hyaluronic acid (HA) technology. The HA component of Tactoset makes the product highly flowable, easily injectable and able to interdigitate trabecular bone architecture with overall improved handling characteristics compared to competitive products. Once injected, Tactoset hardens and mimics the properties of normal trabecular bone initially and remodels into healthy bone over time.
Since its launch in 4Q19, Tactoset has been used for the treatment of bone voids, insufficiency fractures and other skeletal defects, often performed in an outpatient surgical setting. With this expanded indication, surgeons can now use Tactoset in situations where augmenting hardware and/or bone fragments due to insufficient bone quality may be beneficial to their patients. Tactoset has been shown to increase the pull-out strength of a screw-in suture anchor two-fold1 in an osteoporotic bone model when augmented with the cured Tactoset compared to a suture anchor alone.
Anika expects to begin actively marketing the expanded indication for Tactoset for hardware augmentation in October 2021.
Source: Anika Therapeutics, Inc.

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.