
 Copy to clipboard
Copy to clipboard 
3D-Side received another FDA 510(k) clearance for its pre-operative planning software, Customize. This latest clearance allows physicians to use the processing software to pre-plan shoulder replacement surgery.
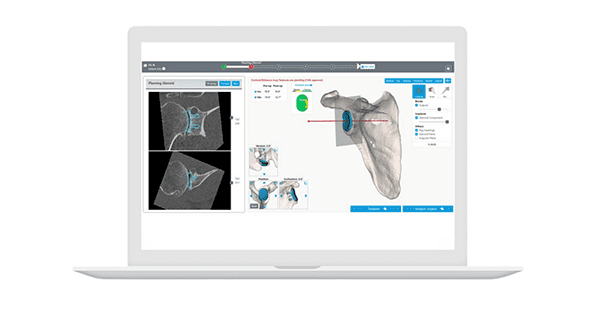
Customize is intended for use during the preparation of shoulder arthroplasties for adult patients only. It displays surgical treatment options that are created based on 3D CAD files generated from multi-slice DICOM data from a CT scanner, and uses Artificial Intelligence during the segmentation process.
The system is a web-based platform where the physician can review CAD files in 3D and modify the position and orientation of the different 3D objects. Customize includes an implant library with 3D digital representations of various implant models so that ideal implant positioning and sizing can be achieved based on the physician’s input.
After physician approval, the treatment plan is saved on the server and can be used as a reference during surgery.
The first partner being onboarded to this new pre-operative planning software is Catalyst OrthoScience, with its platform Archer™ 3D Targeting imaging system.
3D-Side plans to submit another 510(k) for ankle joint replacement in the near future.
Source: 3D-Side
3D-Side received another FDA 510(k) clearance for its pre-operative planning software, Customize. This latest clearance allows physicians to use the processing software to pre-plan shoulder replacement surgery.
Customize is intended for use during the preparation of shoulder arthroplasties for adult patients only. It displays surgical treatment...
3D-Side received another FDA 510(k) clearance for its pre-operative planning software, Customize. This latest clearance allows physicians to use the processing software to pre-plan shoulder replacement surgery.
Customize is intended for use during the preparation of shoulder arthroplasties for adult patients only. It displays surgical treatment options that are created based on 3D CAD files generated from multi-slice DICOM data from a CT scanner, and uses Artificial Intelligence during the segmentation process.
The system is a web-based platform where the physician can review CAD files in 3D and modify the position and orientation of the different 3D objects. Customize includes an implant library with 3D digital representations of various implant models so that ideal implant positioning and sizing can be achieved based on the physician’s input.
After physician approval, the treatment plan is saved on the server and can be used as a reference during surgery.
The first partner being onboarded to this new pre-operative planning software is Catalyst OrthoScience, with its platform Archer™ 3D Targeting imaging system.
3D-Side plans to submit another 510(k) for ankle joint replacement in the near future.
Source: 3D-Side

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







