
 Copy to clipboard
Copy to clipboard 
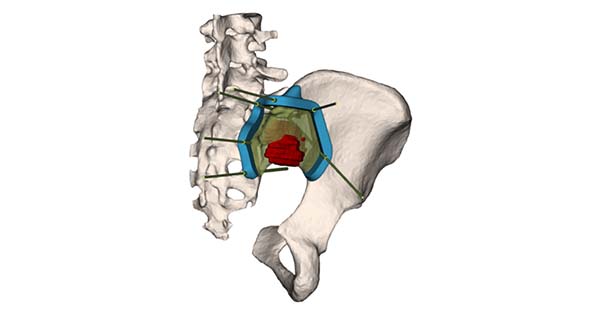
3D-Cut is a patient-matched, additively manufactured single-use surgical instrument (PSI). Based on preoperative planning, the instruments are intended to assist physicians in guiding the marking of bone and guiding surgical instruments in bone tumor resection surgery, excluding joint replacement surgeries. The target locations are femur, tibia and pelvis, including the sacrum.
3D-Cut instruments are designed from patient computed tomography (CT) and magnetic resonance imaging (MRI). The clinician delineates the tumor on the MRI that is merged onto the CT, which produces a 3D model of the bone. A draft resection plan and a PSI design are submitted for evaluation to the treating clinician. Upon surgeon approval, the PSI is produced using additive manufacturing.
This represents the company’s first FDA 510(k) clearance, and validates the Customize software for planning communication and the 3D-printing platform for manufacturing instruments. 3D-Side plans to submit several 510(k)s in the near future for joint arthroplasty to serve its customers.
“This product is a game-changer in the treatment of bone tumors. It gives the surgeons the ability to optimally plan the tumor resection. During the surgery, the 3D-printed instrument accurately transfers the planning to safely remove the bone tumor. The 500 patients treated so far in Europe demonstrate that 3D-Cut actually saves lives,” said Laurent Paul, Ph.D., MBA, Co-CEO of 3D-Side.
Source: 3D-Side
3D-Cut is a patient-matched, additively manufactured single-use surgical instrument (PSI). Based on preoperative planning, the instruments are intended to assist physicians in guiding the marking of bone and guiding surgical instruments in bone tumor resection surgery, excluding joint replacement surgeries. The target locations are femur, tibia...
3D-Cut is a patient-matched, additively manufactured single-use surgical instrument (PSI). Based on preoperative planning, the instruments are intended to assist physicians in guiding the marking of bone and guiding surgical instruments in bone tumor resection surgery, excluding joint replacement surgeries. The target locations are femur, tibia and pelvis, including the sacrum.
3D-Cut instruments are designed from patient computed tomography (CT) and magnetic resonance imaging (MRI). The clinician delineates the tumor on the MRI that is merged onto the CT, which produces a 3D model of the bone. A draft resection plan and a PSI design are submitted for evaluation to the treating clinician. Upon surgeon approval, the PSI is produced using additive manufacturing.
This represents the company’s first FDA 510(k) clearance, and validates the Customize software for planning communication and the 3D-printing platform for manufacturing instruments. 3D-Side plans to submit several 510(k)s in the near future for joint arthroplasty to serve its customers.
“This product is a game-changer in the treatment of bone tumors. It gives the surgeons the ability to optimally plan the tumor resection. During the surgery, the 3D-printed instrument accurately transfers the planning to safely remove the bone tumor. The 500 patients treated so far in Europe demonstrate that 3D-Cut actually saves lives,” said Laurent Paul, Ph.D., MBA, Co-CEO of 3D-Side.
Source: 3D-Side

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







