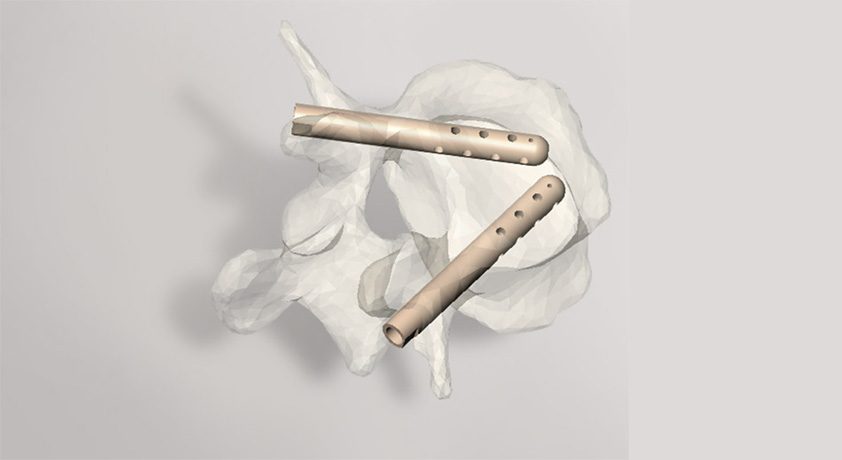
Stryker acquired Vexim, developer of the SpineJack® expandable vertebral compression fracture (VCF) implant, for ~US $191.0MM. The transaction is expected to close in 4Q17. For 2016, Vexim posted revenue of €18.5MM (~$22.0MM), +33% vs. 2015.
Vexim’s portfolio complements Stryker’s Interventional Spine division, which addresses vertebral augmentation, vertebroplasty and radiofrequency ablation procedures as well as diagnostic decompression tools for contained disc herniations. Stryker will gain access to Vexim’s direct salesforce in France, Germany, Spain and Italy, and an international distribution network in Eastern Europe, the Middle East, Latin America and Asia. Vexim will file for FDA 510(k) clearance of SpineJack in 2018, for marketing in the U.S. The product may overtake Stryker’s AVAflex® vertebral augmentation product, acquired from BD in 2Q16.
SpineJack comprises three titanium implants of differing diameters that address 95% of VCFs and all patient morphologies. The 30-minute, minimally invasive x-ray guided procedure is intended to support patient discharge shortly after surgery.
In the overall spine hardware market, ORTHOWORLD estimates place Stryker at #4 among the top five companies ranked by segment revenue—between NuVasive and Globus Medical. Our estimates for Stryker’s 2016 spine revenue of $712.5MM grew by 1.7% vs. 2015, though at 1H17, spine revenue of $172.3MM was down -3.1% vs. 1H16. In the 2Q17 earnings call, leadership noted an ongoing recovery in U.S. spine revenue from the impact of early-2017 supply issues, partially offset by demand for 3D-printed products, with a number of projects in the R&D queue.
Sources: Stryker Corporation; Vexim; ORTHOWORLD estimates
Stryker acquired Vexim, developer of the SpineJack® expandable vertebral compression fracture (VCF) implant, for ~US $191.0MM. The transaction is expected to close in 4Q17. For 2016, Vexim posted revenue of €18.5MM (~$22.0MM), +33% vs. 2015.
Vexim's portfolio complements Stryker's Interventional Spine division, which addresses vertebral...
Stryker acquired Vexim, developer of the SpineJack® expandable vertebral compression fracture (VCF) implant, for ~US $191.0MM. The transaction is expected to close in 4Q17. For 2016, Vexim posted revenue of €18.5MM (~$22.0MM), +33% vs. 2015.
Vexim’s portfolio complements Stryker’s Interventional Spine division, which addresses vertebral augmentation, vertebroplasty and radiofrequency ablation procedures as well as diagnostic decompression tools for contained disc herniations. Stryker will gain access to Vexim’s direct salesforce in France, Germany, Spain and Italy, and an international distribution network in Eastern Europe, the Middle East, Latin America and Asia. Vexim will file for FDA 510(k) clearance of SpineJack in 2018, for marketing in the U.S. The product may overtake Stryker’s AVAflex® vertebral augmentation product, acquired from BD in 2Q16.
SpineJack comprises three titanium implants of differing diameters that address 95% of VCFs and all patient morphologies. The 30-minute, minimally invasive x-ray guided procedure is intended to support patient discharge shortly after surgery.
In the overall spine hardware market, ORTHOWORLD estimates place Stryker at #4 among the top five companies ranked by segment revenue—between NuVasive and Globus Medical. Our estimates for Stryker’s 2016 spine revenue of $712.5MM grew by 1.7% vs. 2015, though at 1H17, spine revenue of $172.3MM was down -3.1% vs. 1H16. In the 2Q17 earnings call, leadership noted an ongoing recovery in U.S. spine revenue from the impact of early-2017 supply issues, partially offset by demand for 3D-printed products, with a number of projects in the R&D queue.
Sources: Stryker Corporation; Vexim; ORTHOWORLD estimates


You are out of free articles for this month
Subscribe as a Guest for $0 and unlock a total of 5 articles per month.
You are out of five articles for this month
Subscribe as an Executive Member for access to unlimited articles, THE ORTHOPAEDIC INDUSTRY ANNUAL REPORT and more.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.