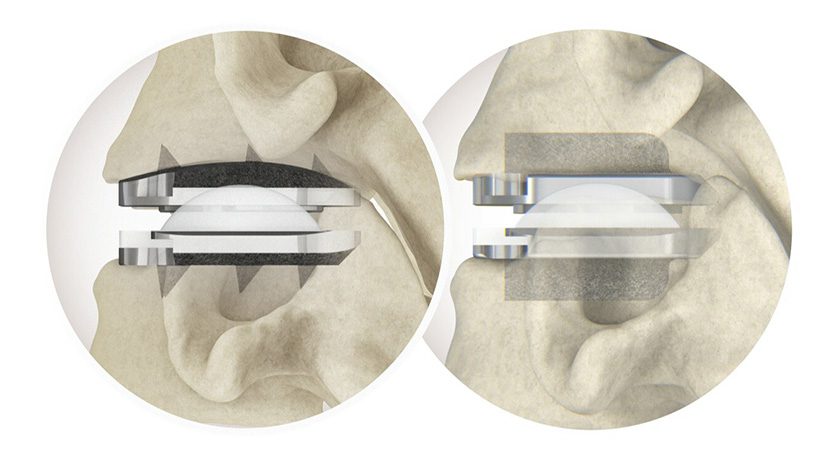
HAPPE Spine has formed to develop and commercialize a material technology that allows for the construction of partial or fully porous hydroxyapatite-reinforced polyetheretherketone (PEEK) implants.
The company’s name derives from HydroxyApatite Porous PEEK. It was formed by Genesis Innovation Group with investments from cultivate(MD) Capital Funds I and II, and is based on the work of Ryan K. Roeder, Ph.D. and researchers from the University of Notre Dame that began over 10 years ago. HA whisker reinforced PEEK with variable porosity can be used to create spinal interbody implants, as well as other potential applications throughout the body. HAPPE will evaluate additional device applications as part of its product strategy.
Genesis Innovation Group and cultivate(MD) Capital Funds will remain partnered with the developers at HAPPE, all of whom will continue as owners, investors and advisors.
As the PEEK vs. titanium conversations continue, variations in PEEK technologies continue to emerge. Earlier this week, Vallum received FDA 510(k) clearance to market a PEEK interbody fusion device with a PEEKplus® nanotextured surface created with proprietary Accelerated Neutral Atom Beam technology.
Sources: Genesis Innovation Group, LLC; ORTHOWORLD Inc.
HAPPE Spine has formed to develop and commercialize a material technology that allows for the construction of partial or fully porous hydroxyapatite-reinforced polyetheretherketone (PEEK) implants.
The company's name derives from HydroxyApatite Porous PEEK. It was formed by Genesis Innovation Group with investments from...
HAPPE Spine has formed to develop and commercialize a material technology that allows for the construction of partial or fully porous hydroxyapatite-reinforced polyetheretherketone (PEEK) implants.
The company’s name derives from HydroxyApatite Porous PEEK. It was formed by Genesis Innovation Group with investments from cultivate(MD) Capital Funds I and II, and is based on the work of Ryan K. Roeder, Ph.D. and researchers from the University of Notre Dame that began over 10 years ago. HA whisker reinforced PEEK with variable porosity can be used to create spinal interbody implants, as well as other potential applications throughout the body. HAPPE will evaluate additional device applications as part of its product strategy.
Genesis Innovation Group and cultivate(MD) Capital Funds will remain partnered with the developers at HAPPE, all of whom will continue as owners, investors and advisors.
As the PEEK vs. titanium conversations continue, variations in PEEK technologies continue to emerge. Earlier this week, Vallum received FDA 510(k) clearance to market a PEEK interbody fusion device with a PEEKplus® nanotextured surface created with proprietary Accelerated Neutral Atom Beam technology.
Sources: Genesis Innovation Group, LLC; ORTHOWORLD Inc.


You are out of free articles for this month
Subscribe as a Guest for $0 and unlock a total of 5 articles per month.
You are out of five articles for this month
Subscribe as an Executive Member for access to unlimited articles, THE ORTHOPAEDIC INDUSTRY ANNUAL REPORT and more.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.