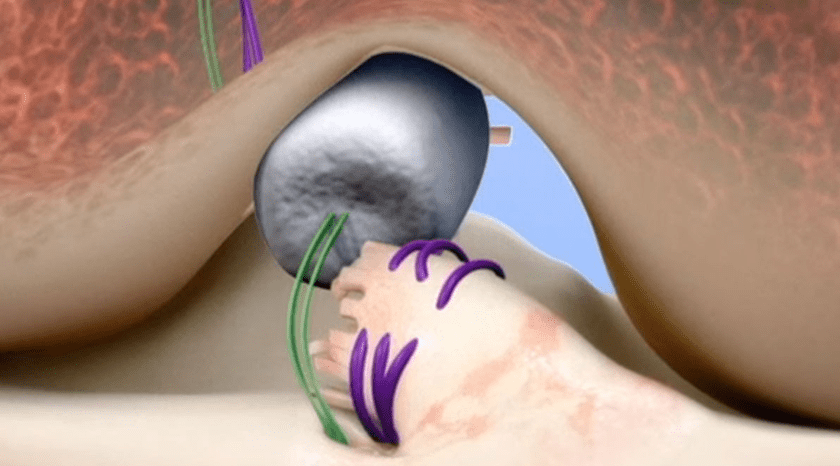
Active Implants completed the first US $10MM tranche of a $40MM Class D Units financing. Funds will support two ongoing clinical trials of the NUsurface® Meniscus Implant, a polymer-based investigational treatment for persistent knee pain following medial meniscus surgery.
Enrollment for the SUN and VENUS trials is expected to be complete by the end of 2017.
The transaction is led by $30MM from first-time investor LS Health Science Partners, with another $10MM to come from existing investors.
Active Implants is exhibiting at AAOS 2017 in Booth #5504.
Sources: Active Implants LLC, ORTHOWORLD Inc.
Active Implants completed the first US $10MM tranche of a $40MM Class D Units financing. Funds will support two ongoing clinical trials of the NUsurface® Meniscus Implant, a polymer-based investigational treatment for persistent knee pain following medial meniscus surgery.
Enrollment for the SUN and VENUS trials is expected to be complete by...
Active Implants completed the first US $10MM tranche of a $40MM Class D Units financing. Funds will support two ongoing clinical trials of the NUsurface® Meniscus Implant, a polymer-based investigational treatment for persistent knee pain following medial meniscus surgery.
Enrollment for the SUN and VENUS trials is expected to be complete by the end of 2017.
The transaction is led by $30MM from first-time investor LS Health Science Partners, with another $10MM to come from existing investors.
Active Implants is exhibiting at AAOS 2017 in Booth #5504.
Sources: Active Implants LLC, ORTHOWORLD Inc.


You are out of free articles for this month
Subscribe as a Guest for $0 and unlock a total of 5 articles per month.
You are out of five articles for this month
Subscribe as an Executive Member for access to unlimited articles, THE ORTHOPAEDIC INDUSTRY ANNUAL REPORT and more.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.